Key Takeaways
- Plasma treatment is widely used in medical manufacturing to improve coating adhesion on polymer tubing such as PET.
- Traditional verification methods like dyne inks are subjective, destructive, and non-quantifiable.
- Measuring water contact angle (WCA) provides objective, non-destructive surface data that correlates directly to coating performance.
- Integrating surface data into BConnect allows manufacturers to track, verify, and audit plasma treatment effectiveness in real time.
- Consistent surface quality ensures uniform coatings, patient safety, and regulatory confidence.
Quality at the Heart of Medical Manufacturing
When manufacturing products that interact directly with the human body, precision isn’t optional, it’s a requirement. From surgical tools to catheters, every surface must function reliably, safely, and predictably. In this environment, process monitoring and surface quality control are not just technical steps, they are essential components of patient safety.
A leading medical tubing manufacturer specializing in catheters faced a recurring challenge: verifying that their plasma treatment step effectively prepared PET (polyethylene terephthalate) tubing before applying a PTFE (polytetrafluoroethylene) coating. This coating provided the lubricity needed for catheters to glide safely through delicate anatomical pathways.
But without a precise way to confirm that the plasma treatment achieved the required surface energy for adhesion, the manufacturer risked coating defects, delamination, and failed product performance.
The Limits of Dyne Ink in a Regulated Environment
Historically, the manufacturer relied on dyne inks to assess surface preparation. While common in R&D labs, dyne inks posed serious limitations on the production floor:
- They are destructive, leaving residue or damaging delicate surfaces.
- The test is subjective, dependent on operator interpretation.
- The results are not quantifiable, providing no reliable traceability.
- Dyne solutions are chemical hazards, complicating use in cleanroom environments.
In short, dyne testing could not provide the objective, in-line verification required by modern medical-device quality systems and regulatory frameworks like ISO 13485 or FDA 21 CFR Part 820.
Objective Surface Verification: Measuring Plasma Treatment Effectiveness
To move beyond guesswork, the manufacturer partnered with Brighton Science to evaluate a non-destructive, data-driven approach.
A Brighton Science sales engineer demonstrated how the Surface Analyst™ measures water contact angle (WCA) directly on production parts with no dyes, no solvents, and no surface damage. WCA provides a direct, quantifiable measure of a material’s surface energy, showing exactly how effectively the plasma treatment activated the polymer surface.
Brighton also supplied a catheter inspection fixture designed to hold tubing of various diameters securely during measurement, ensuring consistency and repeatability.

With this setup, the manufacturer could measure each batch—or even each part—right on the factory floor, verifying that plasma-treated surfaces were ready for coating before the next step in production.
From Surface Data to Process Confidence with BConnect
Beyond immediate verification, the Surface Analyst integrates seamlessly with BConnect, Brighton’s Surface Intelligence platform. Each WCA measurement can be automatically captured, stored, and analyzed, creating a traceable digital record of surface condition for every product run.
Through BConnect, the manufacturer could:
- Monitor plasma process stability over time.
- Identify deviations before defects occur.
- Correlate surface data with coating performance outcomes.
- Provide audit-ready documentation for customers and regulators.
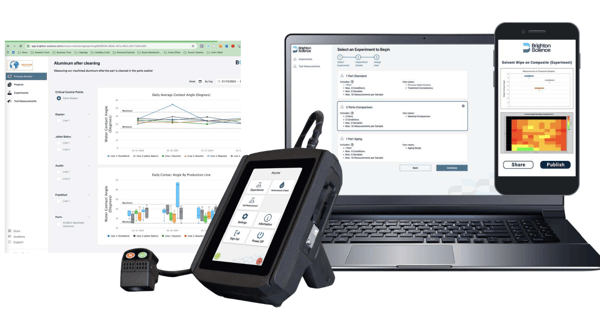
This transformation turned surface verification from a manual inspection task into a real-time quality control process embedded within manufacturing data systems.
Results: Reliable Coatings and Regulatory Assurance
By replacing dyne testing with quantitative surface measurement, the manufacturer achieved:
- Consistent coating adhesion and improved product reliability.
- Reduced rework and scrap, cutting material and labor waste.
- Enhanced compliance documentation, simplifying audits and validations.
- Greater confidence in every catheter that left the facility.
Ultimately, the adoption of objective surface verification ensured that each product performed safely and predictably in the field, protecting patients while streamlining operations.
Surface Intelligence in Medical Manufacturing
Medical device manufacturers operate under intense scrutiny. Every material change, treatment, or coating must be validated and documented. Surface Intelligence bridges the gap between process design and production execution.
By capturing surface data through the Surface Analyst and managing it in BConnect, manufacturers gain:
- Real-time visibility into critical surface control points.
- Digital traceability across multiple facilities and suppliers.
- The ability to proactively optimize adhesion-related processes before failures occur.
Next Steps
To learn more about verifying plasma treatment and creating more efficient, traceable coating processes, download our eBook — “How to Streamline Process Design to Production for Medical Devices.”
See how BConnect powers traceable surface verification in real time. Learn more about BConnect →
FAQ
Q: Why is plasma treatment necessary for PET tubing?
Plasma treatment modifies the surface chemistry of low-energy materials like PET, introducing polar functional groups that improve wettability and enable strong coating adhesion.
Q: How does water contact angle measurement verify plasma treatment?
WCA quantifies the interaction between water and a surface. A lower angle indicates higher surface energy, confirming that the plasma has effectively activated the surface for coating.
Q: Why is BConnect important for regulated manufacturing?
BConnect provides digital traceability, analytics, and centralized data storage, helping manufacturers demonstrate process control and compliance with FDA and ISO standards.
Q: Can this method be applied to other medical components besides tubing?
Yes. The same principles apply to any plasma-treated or coated polymer device, including films, housings, and implantable parts.
It’s fast. It’s accurate. It’s non-destructive. It’s the Automated Surface Analyst.


