Key Takeaways
- Surface tension describes the molecular forces that determine how liquids interact with solids and directly affects adhesion, coating, and wetting performance.
- Measuring surface tension provides predictive insight into product reliability and process control.
- Dynamic surface tension measurement enables real-time optimization of formulations containing surfactants, coatings, or adhesives.
- Understanding surface tension in relation to surface energy (measured through water contact angle) unlocks full control over bonding performance.
- Data intelligence platforms like BConnect allow manufacturers to connect these measurements to process analytics for continuous improvement.
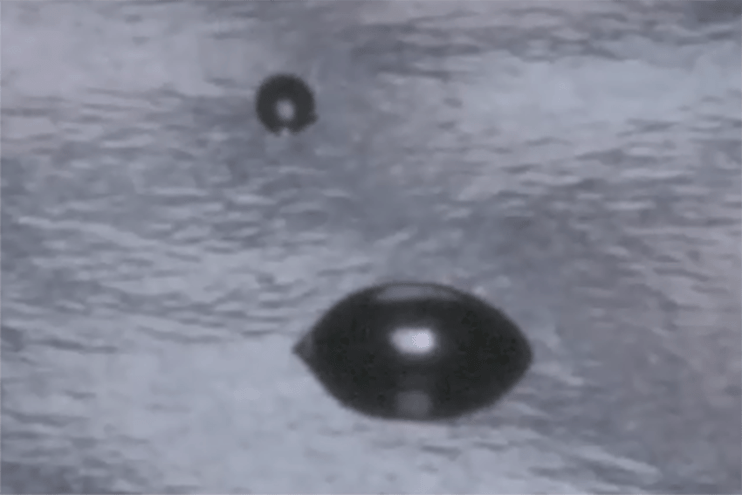
The surface of a liquid might appear perfectly smooth, but at the molecular level, it’s a dense network of cohesive forces. Surface tension is the measurable attraction between molecules at the liquid’s surface and is a property that controls how liquids spread, bead, and bond to other materials.
In manufacturing, those invisible forces determine the reliability of every adhesive, coating, and sealant application. If you can measure them, you can manage them. And if you can manage them, you can prevent failure.
Why Measuring Surface Tension Matters
In adhesion-driven industries such as aerospace, automotive, medical device, and electronics manufacturing, quality and yield depend on controlling molecular-level interactions. Surface tension defines how strongly molecules within a liquid attract one another, while surface energy defines how much a solid surface attracts or repels that liquid. Together, they determine whether a coating spreads evenly, a sealant holds, or a bond forms.
Without quantitative data about these forces, manufacturing processes operate on assumptions rather than evidence. Measuring surface tension provides the first data point in understanding interfacial behavior—the “invisible handshake” that governs every successful bond.
Tip from a Surface Scientist:
Even when materials look clean, molecular-level contaminants can drastically alter surface behavior. Visual inspection alone can’t verify surface readiness for bonding or coating.
Struggling with adhesion failures? Been there. Solved that.
Factors That Affect Surface Tension
Temperature, chemical composition, and molecular structure all influence surface tension, but chemical composition dominates. Temperature changes may slightly reduce surface tension, but it’s the chemistry of the molecules at the top few nanometers that truly controls performance.
Homogeneous liquids (like pure water) have consistent molecular interactions, while blended or multi-component systems (such as inks, paints, or polymer melts) display complex surface behavior. These interactions aren’t simply additive; the way molecules orient themselves at the interface determines the actual surface tension.
Dynamic Surface Tension and Process Control
While surface tension is a physical property, it’s not static. It changes over time as molecules migrate and reorient at the surface. Dynamic surface tension measurement tracks these changes and reveals how fast surfactants, wetting agents, or other additives reach the surface and affect performance.
For example, adding a small amount of surfactant to a liquid might not immediately alter surface tension. But as molecules diffuse to the surface, the measured value drops, changing how the liquid wets or spreads. Understanding this time dependence allows formulators to fine-tune performance under real manufacturing conditions.
Tip from a Surface Scientist:
If your process involves surfactants or wetting agents, monitor dynamic surface tension. Static values can miss critical time-dependent effects that impact coating uniformity and bond strength.
Methods for Measuring Surface Tension
Several techniques accurately measure the surface tension of liquids:- Du Noüy ring and Wilhelmy plate tensiometers measure the force required to detach a ring or plate from the liquid surface.
- Maximum bubble pressure methods evaluate surface tension through pressure differentials as air bubbles form.
However, while a liquid’s surface tension can be directly measured, a solid’s surface energy cannot. Instead, manufacturers rely on correlated measurements, such as water contact angle, to infer the solid’s chemical readiness to bond.
From Surface Tension to Surface Energy
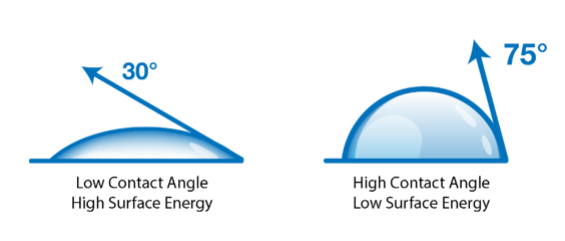
When a liquid droplet contacts a solid surface, it either beads up or spreads out depending on the balance between surface tension and surface energy. Measuring this interaction through contact angle analysis provides a quantitative indicator of surface cleanliness, treatment level, and chemical functionality.
By combining surface tension and contact angle data, manufacturers gain full control of both sides of the bonding equation—the chemistry of the liquid and the condition of the solid.
Tip from a Surface Scientist:
Water contact angle is the most practical, repeatable way to evaluate the surface energy of solids in production environments. A lower contact angle generally indicates higher surface energy and stronger adhesion potential.
Connecting Measurements to Manufacturing Intelligence
Surface tension and surface energy data are only as valuable as the insights they generate. With platforms like BConnect, these measurements become part of a connected quality ecosystem, linking materials, treatments, and process parameters in real time.
By capturing and analyzing surface data across multiple production stages, manufacturers can:
- Identify Critical Control Points (CCPs) where adhesion failures originate.
- Correlate surface condition with downstream yield or warranty data.
- Automate verification of surface preparation, treatment, and cleanliness.
This closed-loop visibility transforms surface science from a laboratory metric into a production control variable, helping teams predict and prevent adhesion-related defects before they occur.
In manufacturing, the unseen molecular forces at the surface determine product success. By measuring, understanding, and connecting those forces through data, you can make adhesion predictable and quality measurable.
To explore how data-driven surface control is reshaping product reliability and quality assurance, download our free eBook:
Frequently Asked Questions
What’s the difference between surface tension and surface energy?
Surface tension applies to liquids, and it measures the cohesive forces between molecules. Surface energy applies to solids and it reflects how strongly a surface attracts or repels liquids.
Can surface tension be controlled in manufacturing?
Yes. Formulators can adjust surface tension by altering chemistry, surfactant concentration, or temperature. Real-time dynamic measurements ensure consistency during production.
Why can’t we measure surface energy directly?
Unlike liquids, solids don’t flow, so their surface energy must be inferred through related measurements like water contact angle, which correlates closely with surface chemistry.
How does BConnect improve surface control?
BConnect integrates surface tension and contact angle data into a single digital framework, enabling traceability, trend analysis, and predictive process intelligence for adhesion-critical operations.